News
Fine supports Intellus Compliance Training
FINE has participated in an expert panel about Compliance run by Intellus Worldwide.
Intellus Worldwide is the leading association for healthcare insights and analytics, including global manufacturers and service providers within pharmaceuticals, biotech, medical device and diagnostics.
The podcast featured a number of experts including, Lisa Courtade, Organon, Jessica Santos, Kantar Health, Cedric Degraeuwe, QualWorld and Diego Casaravilla, Fine Research Latin America, and was moderated by Heidi Boyle, Director of Events & Education of Intellus Worldwide
The detailed topics included:
Trends and regulations to watch
How to handle disclosure and international data transfer
How to innovate within the guardrails
Knowledge insights professionals should have to be conversant, and safe
Podcasts aims to help researchers to approach compliance issues in upcoming projects with confidence, and feel more fully prepared to engage with clients or internal stakeholders.
Intellus Members can access the full discussion via the Virtual Learning Center.
Read More
FINE participates in an EphMRA panel about COVID-19 impact
June 2021
EphMRA, the European Pharmaceutical Market Research Association kindly invited, Florencia Rojo, Account Manager at FINE to take part of an expert panel about COVID-19 impact on primary research in emerging economies.
The panel was moderated by Stephen Potts, Director at Purdie Pascoe (UK) and while Florencia exposed about Brazil and rest of Latin America, Asebhor Ebhomenye (ABA Healthcare, Nigeria) did the same with Africa, and Johana Ruszczak-Zbikowska (IQVIA Poland) presented her views on the situation in Eastern Europe.
While each region have its unique challenges, all participants agreed on how the accelerated growth of digital alternatives has become a viable and successful solution for most market insights data collection requirements.
For more details about EphMRA webinars please check https://www.ephmra.org/events-training/
Read MoreThe future is here: Esomar Director General, Finn Raben, interviews Fine´s CEO
June 2021
Esomar, the largest world association of research professionals, is running a few interviews with leading researchers all over the world in a series of episodes called “THE FUTURE IS HERE”.
The proposal is to “Join ESOMAR’s Director General as he explores with business leaders from around the world how the future of business is being shaped by COVID-19 and how they prepare for a post-COVID-19 world. How are companies preparing for the digital transformation?”
The first episodes included interviews with Joris Hiusmanm, (Managing Partner of SKIM), Chris Farquhar (Managing Partner of Cimigo) and Joy Uyanwune (Global Marketing Director at Decision Support).
On June 4th, Esomar released EPISODE 4, in which Finn Raben, ESOMAR´s Director General interviewed Diego Casaravilla, CEO of FINE.
The discussion covered a number of topics including:
– How long will it be before we get “back” to pre-Covid levels of business?
– Change in client demands and expectations
– Meaning of the “age of platforms” in our industry
– Changes to team and business work practices
– Advice to researchers and young entrepreneurs when looking at LatAm
Check the full interview in YouTube
Read MoreLatAm HCPs trust that vaccines will put an end to the pandemic putting more confidence on those that provide locally a credible evidence story
CLICK HERE TO GET A FREE COPY OF THE REPORT (ENGLISH)
On March 31, 2021 during the ESOMAR event Transformation and Trust, Fine released the latest results of COVID-19 Tracking study.
This is part of an initiative launched in March 2020 and that has involved responses of over 13,000 doctors in 16 countries.
The report of the 6th wave which took place in March 2021 was based on a sample of 2451 doctors from Brazil, Mexico, Colombia, Argentina and Chile and was focused in the vaccines.
The survey firstly details an evolution of HCPs main challenges showing that the drop in the number of patients seen, although relevant for many and still representing in average a 28% fall compared to pre-pandemic levels, is no longer the main challenge for most physicians as it was during 2020. In fact work overload surges as a growing challenge.
Their main current concern remains the risk of infection in the context of interaction between physicians and patients. This concern is consistent with the fact that almost 60% have been in contact with infected patients in the past week and that the incidence of COVID-19 in the medical population is high and many times the proportion of the general population (by the survey date 21% of the participating HCPs were diagnosed with COVID), especially in Mexico where 30% of professionals report having been infected.
At the time of the survey (March 9-10), 73% of physicians in the region reported having received at least one COVID-19 vaccine dose and 43% both doses. This level of vaccination coverage of professionals with at least one dose, was higher in Chile, Brazil and Argentina, while it barely covered about half of the professionals in Colombia and Mexico.
Physicians in LatAm are mostly confident that vaccines will put an end to the pandemic, considering that the evidence supporting them is solid and that the quality of the vaccines available in the region is similar to that of Europe and the United States.
In addition, half of physicians have strong confidence in the safety of vaccines, and the other half believe that although they are not completely safe, vaccination is still more beneficial for the population. Consequently 95% of physicians recommend vaccination to their patients.
Regarding the specific vaccine options, Pfizer’s vaccine clearly leads regionally followed by Astra Zeneca, Sputnik (strong in Argentina and Mexico, but marginal in Brazil and Chile), Moderna, Janssen and Sinovac.
The main drivers that generate trust for the different vaccines were those that contributed to build locally a credible evidence story. In other words the most trusted ones are those that show good results in phase three trials regarding safety, general effectiveness and effectiveness to reduce severe cases and mortality.
The ability to deal with new varieties seems to be les relevant, but this is likely to be affected by the fact that no vaccine is “owning” this attribute which highlights a potential risk scenario.
The made-in-label prestige and the manufacturer reputation are relevant but not always decisive and for instance Sputnik in Argentina or Sinovac in Chile get high trust levels even when they are rated comparatively lower in these prestige variables.
While trust in vaccines ranks high in general, confidence in governments and national vaccination plans is significantly lower.
Only 20% of physicians strongly or somewhat agree with the health policies in place. They collectively estimate that about half of the population would be vaccinated by the end of 2021 and that it would take about 20 months to bring the pandemic under control. The WHO COVAX program does not raise many expectations either.
Chilean physicians are significantly more optimistic than their peers in the region in their evaluation of health policy management, especially with regard to vaccination plans in which they are almost unanimously confident. They estimate that in 11 months the pandemic will be definitively under control in the country (is about half the time estimated by their peers in the region) and that 77% of the population will be vaccinated by the end of the year.
The survey also valued the role of other stakeholders, and stands out that HCPs values a positive role for the pharmaceutical industry in the context of the pandemic. An industry that traditionally had a more criticized image in polls seems to be revalued and 6 out of 10 doctors state that the development of vaccines has contributed to improving their image of the industry, especially of Pfizer, Astra Zeneca and Jannsen.
Click HERE to learn more about FINE or access other reports
Read More
Fine had an active role at ESOMAR first flagship 2021 event
On March 31st 2021, ESOMAR run its first 2021 flagship event which was called Transformation and Trust.
ESOMAR based in Amsterdam, is a not-for-profit organisation that promotes the value of market, opinion and social research and data analytics. It was created in 1947 and is currently the largest and most renowned organization of this kind in the Insights Industry.
Fine´s CEO Diego Casaravilla, integrated the event advisory board jointly with leading professionals from all around the globe: Omar G. Aragon (PepsiCo, Mexico), Fiona Blades (MESH Experience, UK), Maya Kantak (Disney Parks Experiences and Products, USA), Gigi Leung (Johnson & Johnson Vision, Japan) and Jennifer Lin (Answer Global, Taiwan).
Diego and Omar (Pepsico) chaired the LatAm episode which included presentations of Coca-Cola, Kimberly Clark, Netquest or Mercado Senior.
In one of the sessions, Florencia Rojo (Accounts Director, Fine) presented jointly with Andrey Mendez (Director Southern Cone, Ipsos Healthcare) a summary of the results of the last wave of Fine COVID-19 Tracking Study with focus on how trust is built around vaccines and other key insights. You can watch the presentation given at the Transformation and Trust event (in Spanish) by clicking here.
Read MoreFear of infection during consultation has become a serious health risk
There is no doubt that fear of infection has played a role against the pandemic. Given lack of effective treatments or implementation of trusted and approved vaccines, the social response to the pandemic has been one of the dominant drives that defined the curves of infection in the different societies. Those that due to fear of infection of themselves or their loved ones have shown lhigher risk avoiding attitudes like social distancing, use of protection or limiting social activities were indeed less likely to have got infected. However a recent study run by Fine Research with over 2500 HCPs in LatAm shows that this same fear of infection is also driving a negative effect on health. In this study doctors estimated that before the pandemic roughly 7 out of 10 of their patients were being able to be properly compliant to their prescribed treatment. When asked about their current patient adherence to their treatments now they estimated that this was reduced and only 6 out of 10 are being able to do so. And this is affecting all kind of life threatening diseases such as cardiodological diseases, cancer, diabetes or HIV. What is more worrying is that reasons for non-compliance have also changed abruptly. While in the past the main reasons for low treatment adherence were treatment access restrictions including high cost of medicines, red tape or lack of approval of the best treatment options, now most physicians state that main reason for low treatment compliance is just that patients are afraid of getting infected during the consultation. In the region the same survey shows that the number of patients cared by doctors have dropped in October by 35% compared to pre-pandemic levels, this is indeed a recovery compared to the sharp drop of 61% observed by a previous measurement in May, but still shows that the aggregated effect will have a significant impact on the health of the population. In addition fear is not only affecting those that have a specific diagnosis but also those who are in risk of getting a disease. Doctors tell that they are performing less than half the number of cardiological check ups and about one third of the prostate and breast cancer controls, that they were doing before the pandemic. This obviously is meaning a likely higher number of cardiovascular events or late detected cancers which will likely impact mortality rates. Consistently, when checked by specialty cardiologists, hematologists and oncologists are the most concerned about potential life impact of lack of in person care which could put in risk over half of their patients. In the other extreme, few psychiatrists see a similar risk with the added advantage that they are the ones who mostly perceive virtual consultation as a good fit to their practice in the current context and also in the future. Regarding COVID-19 infections, hospitals are perceived as showing the higher risks, particularly in the hospitalization areas, and also if they are public, and COVID-19 reference centers. In comparison care taking place in doctor’s office, clinics or separated outpatient areas becomes a safer option not only based on HCPs assessment but also on the levels they have witnessed of own or patient infections in the different work settings In summary, the data from this survey with physicians all over Latin America, shows that the healthcare system needs to continue developing protocols for safe care and properly communicate them so that not only the risk of infection is minimized but also patients could gain confidence and get back to meet their doctors to adequately address their own health. Access here for a free download of the full report to check the emerging trends and how these vary among different countries and medical specialties. Resu;lts include details about transformation of the medical practice and relationships of HCPs and the pharma industry among other insights.
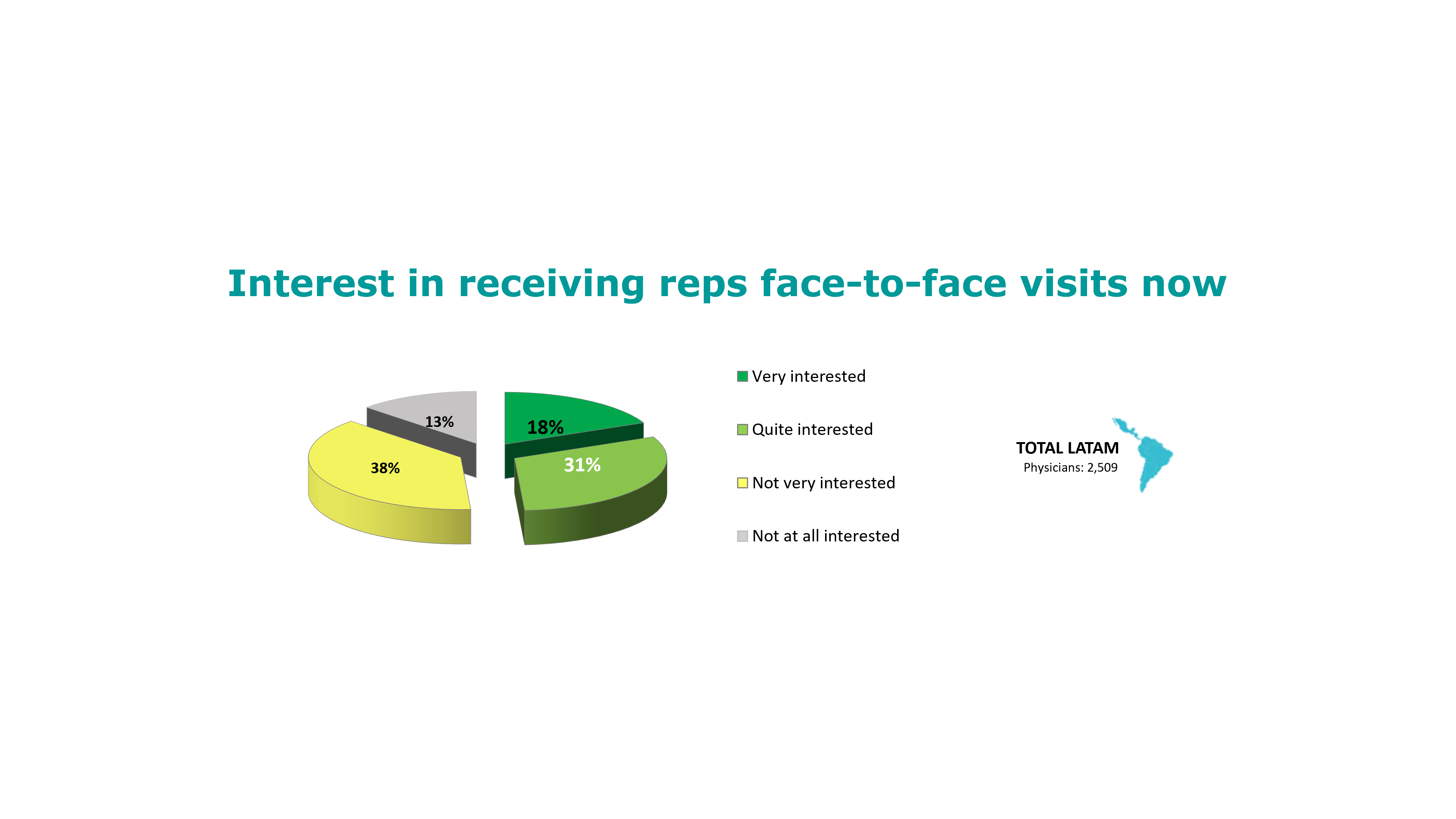
Read MoreHalf of LatAm HCPs interested in f2f rep visits now and 9 out of 10 once pandemic is controlled
Released November 2020
The New landscape for HCPs in Latin America
Changes in Medical Practice & relationships with Pharma Reps
A new report by Fine’s Latin American Healthcare Panel, contains fresh data collected from our ongoing COVID-19 Tracking Study.
This has become a must-read for any researcher or marketer interested in developing optimized communications with physicians in Latin America.
Download this free report to prepare for the future of pharma-HCP interactions ->
By reading this report you will learn latest news such as:
– 4 out of 10 doctors claim to have introduced new therapies into their practice during pandemic.
– LatAm HCPs see recovery in number of patients but still show reduced adherence and very low levels of patient controls. – Half of LatAm doctors are interested in receiving in-person pharma reps visits now, and 9 out of 10 would like to have them once the pandemic is controlled. ACCESS HERE FOR A FREE DOWNLOAD OF THE FULL REPORT to check the emerging trends and how these vary among different countries and medical specialties.
About this initiative
Since March 2020, Fine Research has been running a series of tracking waves around COVID-19 impact on Healthcare Professionals in Latin America, including over 11.000 interviews in 15 different countries.
Results have been widely published in the press and media in several countries as well as in articles and webinars sponsored by Market Research industry forums such as ESOMAR Foundation, ESOMAR, Research News, EphMRA, Brazilian Association of Research Companies, Brazilian Association of Research professionals, and GreenBook.
Read More
Fine distinguished with an Outstanding Achievement in Insight & Research Award for Third Consecutive Year
New York, NY, Oslo, Norway, and London, UK —December 12th, 2021— FORSTA announced the winners of the 2021 AIR (Achievement in Insight and Research) Awards. The annual AIR Awards recognize FORSTA customers committed to innovation and the advancement of the Market Research industry.
We are extremely proud to share that for third consecutive year, Fine Research won an Award in the AIR Project & Innovation category. The company is so far the only LatAm company that has won an AIR award
2021 edition was evaluated by an impressive crew of judges who included Fiona Blades (MESH), Simon Chadwick (CAMBIAR), Shannon Danzy, (WEARERALLY), Natalie Samuel, (COLOUR OF RESEARCH) and Kristin Luck, (SCALEHOUSE) who is currently ESOMAR president.
“I am delighted to congratulate Fine Research as a 2021 Forsta AIR Award winner for their proven excellence in insight and research. Fine Research ’s dedication to provide exceptional guidance and actively deliver significant value to their clients is a credit to their team. The global research and insights industry is driven by the energy of these organizations and we are proud to partner with them as they continue to deliver some of the most innovative research approaches in the industry” commented Kyle Ferguson, CEO at Forsta.
In 2020, we also got very positive comments and the judges said: “Great insights and important work. Fine Research has a great partnership with Save the Children and ESOMAR Foundation. They have approached this in the right way – no incentive. This is an excellent case study!”
Diego Casaravilla, CEO of Fine Research added “We are very sure that these recognitions to our work will be a strong motivation to all and every team member to continue committing to excellence as the most adequate possible response to these uncertain times.”
Read MoreFine Health Panels: Thriving in the Hardest Contexts
August 2020
There is no doubt that the Insights industry is being severely impacted by the COVID-19 pandemic. An ESOMAR study conducted earlier this year forecasts a 22% fall for all research related companies. In Latin America data provided by local organizations such as the Brazilian Association of Researchers suggests the hit might be even harder.
However the impact is not equal to all parties and approaches. While traditional Focus Groups and quantitative f2f are now almost completely out of the radar, digital tools are in much better shape and taking the advantage of an accelerated digital transition, that is likely to remain in the long run after the pandemic.
FINE has been able to witness the resilience of its digital methods in the healthcare space in Latin America. On the one hand, it has been able to run over 10,000 interviews with physicians around COVID-19 without need to push respondents with monetary incentives. This was run from March to July in the peak of the pandemic as described in an article published at the ESOMAR FOUNDATION providing evidence of the impact of COVID-19 on the healthcare profession.
On the other hand, a project conducted by FINE with the support of Toluna and Reckner Healthcare, have shown how digital tools can be used to get independent healthcare evidence in sensitive contexts.
The collected data exposed the dramatic situation in terms of nutrition, drug access and increased epidemic risk in Venezuela, in spite of the complex political situation in that country and the fact many citizens could have a legitimate fear to express their true opinions.
These platforms enabled for an anonymous participation generating evidence that would have been strongly biased if intermediated by an interviewer, as described by our article in Research World.
And this of course does not limit to these specific not for profit projects that could be easily shared since were publicly published. The same is valid in our many business research assignments and in fact at FINE we have seen an increased response rates in our client sponsored projects during the pandemic.
Physicians are showing a larger preference for online tools, and this is happening globally. Recent reports published by EphMRA and Intellus Worldwide confirm that the majority of HCPs are interested in keeping up doing Market Research surveys but strongly prefer a digital approach. Phone is only accepted by a minority and for very short surveys only, while F2F has clearly been left out of the menu.
Of course online engagement requires a long lasting relationship, a trusted brand name, transparent and solid procedures in full compliance with the industry rules and giving value to the respondents.
FINE panelists are getting exclusive content such as access to own surveys on the medical profession, evidence-based medicine resource with free access to DYNAMED, or involvement in not-for-profit campaigns to support renowned charities. This unique approach makes FINE, the leading healthcare research panel in Latin America in terms of volumes, timing, deliverability and quality.
Please feel to reach out to see how we can support your data collection needs with HCPs using our digital tools, writing to info@fine-research.com
Read More